Rational integration of spatial confinement and polysulfide conversion catalysts for high sulfur loading lithium–sulfur batteries†
Abstract
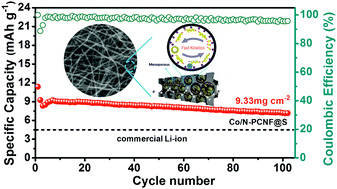
Spatial confinement is a desirable successful strategy to trap sulfur within its porous host and has been widely applied in lithium–sulfur (Li–S) batteries. However, physical confinement alone is currently not enough to reduce the lithium polysulfide (Li2Sn, 4 ≤ n ≤ 8, LIPSs) shuttle effect with sluggish LIPS-dissolving kinetics. In this work, we have integrated spatial confinement with a polar catalyst, and designed a three-dimensional (3D) interconnected, Co decorated and N doped porous carbon nanofiber (Co/N-PCNF) network. This Co/N-PCNF film serves as a freestanding host for sulfur trapping, which could effectively facilitate the infiltration of electrolyte and electron transport. In addition, the polar Co species possess strong chemisorption with LIPSs, catalyzing their reaction kinetics as well. As a result of this rational design and integration, the Co/N-PCNF@S cathode with a sulfur loading of 2 mg cm−2 exhibits a high initial discharge capacity of 878 mA h g−1 at 1C, and maintains a discharge capacity of 728 mA h g−1 after 200 cycles. Even with high sulfur loading of 9.33 mg cm−2, the cathode still keeps a stable areal capacity of 7.16 mA h cm−2 at 0.2C after 100 cycles, which is much higher than the current areal capacity (4 mA h cm−2) of commercialized lithium-ion batteries (LIBs). This rational design may provide a new approach for future development of high-density Li–S batteries with high sulfur loading.


 Please wait while we load your content...
Please wait while we load your content...