Role of the anatase/TiO2(B) heterointerface for ultrastable high-rate lithium and sodium energy storage performance†
Abstract
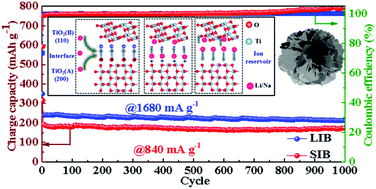
This paper is dedicated to elucidating the role of the anatase/TiO2(B) heterointerface, which functions as an ‘ion reservoir’ for dominant pseudocapacitance, for ultrastable high-rate energy storage in both Li-ion and Na-ion batteries (LIBs, SIBs). Dual-phase nanosheets are in situ assembled to form anatase/TiO2(B) nanoflower-shaped anodes via a facile hydrothermal and thermolysis process. The abundant oxygen vacancies on the ultrathin nanosheets favor pseudocapacitive behaviors and fast ionic/electronic transport during Li+/Na+ insertion/extraction cycles. The density functional theory calculations combined with ab initio molecular dynamics simulations corroborate the important role of the anatase/TiO2(B) heterointerface in promoting electrochemical kinetics. The heterointerface has much lower adsorption energies of Li+/Na+ than in each phase acting alone, and the presence of an internal electric field with a high ionic concentration at the interface ameliorates charge transport. Therefore, the dual-phase anodes deliver ultrastable electrochemical performance with high specific capacities of 193 and 112 mA h g−1 at an exceptionally fast 20 C in LIBs and SIBs, respectively. Their cycling stability is equally remarkable, sustaining reversible capacities of 212 mA h g−1 at 10 C and 173 mA h g−1 at 5 C after 1000 cycles, respectively. These new findings may help rationally design high-performance multi-functional anodes for next-generation metal-ion batteries.


 Please wait while we load your content...
Please wait while we load your content...