Production of Fe nanoparticles from γ-Fe2O3 by high-pressure hydrogen reduction
Abstract
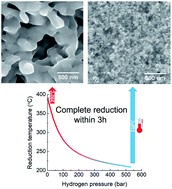
In this work, the reduction of iron oxide γ-Fe2O3 nanoparticles by hydrogen at high pressures is studied. Increasing the hydrogen pressure enables reduction of γ-Fe2O3 to α-Fe at significantly lower temperatures. At low pressures, a temperature of 390 °C is necessary whereas at 530 bar complete reduction can be realized at temperatures as low as 210 °C. This leads to significant improvement in the final particle morphology, maintaining high surface-to-volume ratio of the nanoparticles with an average size of 47 ± 5 nm which is close to that of the precursor γ-Fe2O3. Neck formation, coalescence and growth during reduction can be significantly suppressed. Investigations of magnetic properties show that saturation magnetization of the reduced α-Fe nanoparticles decreases with particle size from 209 A m2 kg−1 at 390 °C reduction temperature to 204 A m2 kg−1 at 210 °C. Coercivity for the fine iron particles reaches 0.076 T which exceeds the theoretical anisotropy field. This is attributed to nano-scale surface effects.
- This article is part of the themed collection: Welcome to the community


 Please wait while we load your content...
Please wait while we load your content...