A fluorous biphase drug delivery system triggered by low frequency ultrasound: controlled release from perfluorous discoidal porous silicon particles†
Abstract
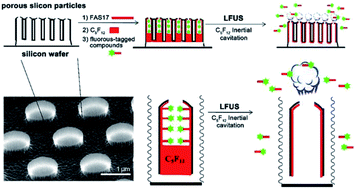
Conventional drug delivery systems face unsatisfactory loading efficiency, poor biological bypass, and uncontrollable release, which are great barriers for improving the treatment of many diseases. Herein, a proof-of-concept of a fluorous biphase drug delivery system (FB-DDS) trigged by low frequency ultrasound (LFUS) is proposed for the first time, where promoted incorporation and stabilization of therapeutic agents in nanocarriers was achieved through fluorine–fluorine interactions, and the encapsulated drugs were controllably released by external sources, resulting in minimized nonspecific toxicity and enhanced therapeutic efficacy. The FB-DDS was constructed from monodisperse, discoidal porous silicon particles (PSP) and was functionalized with 1H,1H,2H,2H-perfluorodecyltrimethoxysilane (FAS17) for loading perfluoropentane (PFP) and fluorinated drugs through fluorine–fluorine interactions. This delivery system was demonstrated by utilizing model compounds including a fluorous-tagged fluorescein and a fluorine containing antibiotic ciprofloxacin. Loading of the model molecules into fluorocarbon-coated carriers was facilitated by fluorous interactions, whereas ejection of the model molecules was promoted by applying LFUS to rapidly evaporate PFP. In the in vitro test, these carriers loaded with fluorine containing ciprofloxacin exhibited excellent antimicrobial activity against Pseudomonas aeruginosa biofilm formation. Overall, this innovative stimulus-responsive fluorous biphase drug delivery system will be a promising candidate for practical applications as well as encouraging further investigation of drug delivery and controlled release strategies.


 Please wait while we load your content...
Please wait while we load your content...