Electrochemical performance of a self-assembled two-dimensional heterostructure of rGO/MoS2/h-BN†
Abstract
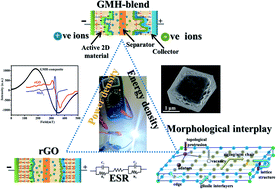
We report the preparation and electrochemical performance evaluation of a two-dimensional (2D) self-assembled heterostructure of graphene oxide (rGO), molybdenum disulphide (MoS2), and hexagonal boron nitride (h-BN). In the present study, the rGO–MoS2–h-BN (GMH) multi-layered GMH heterostructure is fabricated via an in situ chemical route. Based on material analysis, the composite consists of bond conformations of C–B–C, Mo–S, C–N, B–N, and Mo–C, indicating the layered stacks of rGO/h-BN/MoS2. In electrochemical analysis, the composite showed superior performance in the aqueous medium of cobalt sulphate (CoSO4) over other samples. CV measurements, performed over the range 10 to 100 mV s−1, showed a change in specific capacitance (Csp) from 800 to 100 F g−1. GMH showed almost no degradation up to 20 000 cycles @ 100 mV s−1. The calculated Csp, energy density (ED), and power density (PD) are discussed in light of Nyquist, Bode, and Ragone analysis. An equivalent circuit is simulated for the cell and its discrete electronic components are discussed. Due to its larger effective electron diffusion length > 1000 μm, broadly, the composite showed battery-like characteristics, as supported by radical paramagnetic resonance and transport response studies. The symmetric electrodes prepared in one step are facile to fabricate, easy to integrate and involve no pre or post-treatment. They possess superior flat cell character, are cost effective, and are favourable towards practicality at an industrial scale, as demonstrated on the laboratory bench. The details are presented.


 Please wait while we load your content...
Please wait while we load your content...