Preparation of a Ti0.7W0.3O2/TiO2 nanocomposite interfacial photocatalyst and its photocatalytic degradation of phenol pollutants in wastewater†
Abstract
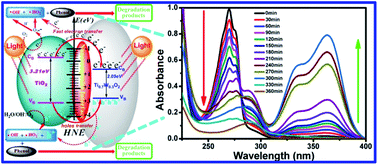
A Ti0.7W0.3O2/TiO2 nanocomposite interfacial photocatalyst was designed and prepared for the photocatalytic degradation of phenol pollutants in wastewater. The detailed properties of the Ti0.7W0.3O2/TiO2 nanocomposite interface (NCI) were analyzed by XRD, SEM, EDX, DRS, UPS and XPS technologies, showing that anatase TiO2 nanospheres (NSs) were uniformly dispersed on the surface of rutile Ti0.7W0.3O2 nanoparticles (NPs) and formed the nanocomposite interface. The DRS and UPS results of 5 wt% Ti0.7W0.3O2/TiO2 NCI indicated a greatly broadened light response range with a wavelength shorter than 527 nm and a shorter band gap energy of 2.37 eV. The conduction band of TiO2 NSs, Ti0.7W0.3O2 NPs and 5 wt% Ti0.7W0.3O2/TiO2 NCI were measured based on the results of the valence band and band gap energy obtained via XPS and DRS, and then the energy level diagram of Ti0.7W0.3O2/TiO2 NCI was proposed. The photocatalytic degradation of phenol at Ti0.7W0.3O2/TiO2 NCI with different loading ratios of Ti0.7W0.3O2 NPs was investigated under optimum conditions (i.e., pH of 4.5, catalyst dosage of 0.45 g L−1 and phenol initial concentration of 95 ppm) under the illumination of ultraviolet visible light. Also, 5 wt% Ti0.7W0.3O2/TiO2 NCI exhibited the highest photocatalytic activity, with the initial rate constant (k) calculated as 0.09111 min−1. After recycling six times, Ti0.7W0.3O2/TiO2 NCI showed good stability and recyclability. The involvement of superoxide radicals in the initial reaction at Ti0.7W0.3O2/TiO2 NCI was evidenced by the use of a terephthalic acid (TA) fluorescent probe. Besides, UV-Vis spectroscopy, UHPLC-MS and GC-MS technologies were used to analyze the main intermediates in the photocatalytic degradation of phenol. The probable photocatalytic degradation mechanism of phenol at Ti0.7W0.3O2/TiO2 NCI was also proposed.
- This article is part of the themed collection: Photocatalysis and Photoelectrochemistry


 Please wait while we load your content...
Please wait while we load your content...