MiR-196-5p involvement in selenium deficiency-induced immune damage via targeting of NFκBIA in the chicken trachea†
Abstract
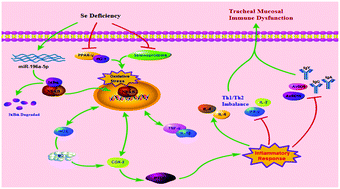
Dietary selenium (Se) deficiency can induce multifarious immune injury in tissues, accompanied by inflammation and a decreased expression of selenoproteins. The results of previous studies indicated that these issues are associated with Se-mediated microRNAs involved in immune regulation, although the specific mechanisms associated with these interactions have not been reported in the trachea of chickens. To explore the effects of Se deficiency in the trachea of chickens and the role of miR-196-5p, we established correlational models of tracheal injury in chickens. One hundred broilers were divided into four groups, including a control group (C group), a Se deficient group (L group), a lipopolysaccharide (LPS)-induced control group (C + LPS group) and a LPS-induced Se deficient group (L + LPS group). Light microscopy observations indicated that the infiltration of inflammatory cells was the major histopathological change caused by Se deficiency. Furthermore, ultrastructural observation of the tracheal epithelium and ciliary showed typical inflammatory signs owing to Se deficiency. We determined the targeting relationship between miR-196-5p and NFκBIA by bioinformatics analysis. In the case of Se deficiency, the changes were detected as follows: 19 selenoproteins showed different degrees of decrease (p < 0.05). Significant inhibition of both antimicrobial peptides and immunoglobulin production were observed (p < 0.05). IκB-α (NFκBIA) expression degraded with the increasing miR-196-5p (p < 0.05), and the NF-κB pathway was activated. Thereafter, we can see a significant increase in the mRNA levels of inflammatory cytokines-related genes (tumor necrosis factor (TNF)-α, inducible nitric oxide synthase (iNOS), cyclooxygenase (COX)-2, prostaglandin E (PTGE), interleukin (IL)-1β, IL-6) and protein expression of NF-κB/iNOS pathway-related genes (NF-κB, iNOS, TNF-α, COX-2) (p < 0.05). The release of IL-2, interferon (IFN)-γ inhibited (p < 0.05) and the secretion of IL-4, IL-6 increased, suggesting the imbalance of Th1/Th2 (Th, helper T cell) cytokines. Compared to the control, the mRNA and protein expression levels of the anti-inflammatory system components with antioxidant activity (PPAR-γ/HO-1) were in an inhibitory state (p < 0.05). Antioxidases (SOD, CAT, GSH-Px) activities were suppressed. The activities of the peroxide markers (MDA, H2O2) were enhanced (p < 0.05). In addition, Se deficiency had a positive effect on the pathological changes of inflammation and the exceptional immunity in LPS-treated groups (p < 0.05). The results confirmed the relationship between miR-196-5p and NFκBIA in chickens, revealing that Se deficiency causes respiratory mucosal immune dysfunction via the miR-196-5p-NFκBIA axis, oxidative stress and inflammation. Moreover, Se deficiency exacerbates the inflammatory damage stimulated by LPS. Our work provides a theoretical basis for the prevention of tracheal injury owing to Se deficiency and can be used as a reference for comparative medicine. Furthermore, the targeted regulation of miR-196-5p and NFκBIA may contribute to the protection of the tracheal mucosa in chickens.


 Please wait while we load your content...
Please wait while we load your content...