A recombinant platform to characterize the role of transmembrane protein hTMEM205 in Pt(ii)-drug resistance and extrusion†
Abstract
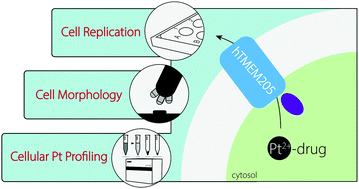
Platinum-coordination complexes are among the most effective chemotherapeutic drugs used in clinics for the treatment of cancer. Despite their efficacy, cancer cells can develop drug resistance leading to treatment failure and relapse. Cellular uptake and extrusion of Pt(II)-complexes mediated by transmembrane proteins are critical in controlling the intracellular concentration of Pt(II)-drugs and in developing pre-target resistance. TMEM205 is a human transmembrane protein (hTMEM205) overexpressed in cancer cells that are resistant to cisplatin, but its molecular function underlying – resistance remains elusive. We developed a low-cost and high-throughput recombinant expression platform coupled to in vivo functional resistance assays to study the molecular mechanism by which the orphan hTMEM205 protects against Pt(II)-complex toxicity. Based on the original observation by the Rosenberg group, which led to the discovery of cisplatin, we performed quantitative analysis of the effects of Pt(II)-coordination complexes on cellular growth and filamentation in E. coli cells expressing hTMEM205. By coupling our methods with Pt quantification and cellular profiling in control and hTMEM205-expressing cells, we demonstrate that hTMEM205 mediates Pt(II)-drug export selectively towards cisplatin and oxaliplatin but not carboplatin. By mutation analysis, we reveal that hTMEM205 recognizes and allows Pt(II)-extrusion by a putative sulfur-based translocation mechanism, thereby resulting in pre-target resistance. Thus, hTMEM205 represents a new potential target that can be exploited to reduce cellular resistance towards Pt(II)-drugs.


 Please wait while we load your content...
Please wait while we load your content...