2,2′-Dipyridyl diselenide (Py2Se2) induces G1 arrest and apoptosis in human lung carcinoma (A549) cells through ROS scavenging and reductive stress†
Abstract
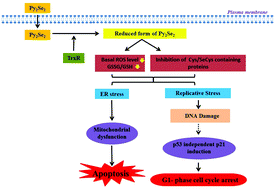
Organo-diselenides are well documented for pro-oxidant effects in tumor cells. However, the present study demonstrated that 2,2′-dipyridyl diselenide (Py2Se2) induced cytotoxicity in human non-small cell lung carcinoma (A549) cells through reductive stress marked by a significant decrease in the basal level of reactive oxygen species and a concurrent decrease in the ratio of oxidised (GSSG) and reduced (GSH) glutathione. The IC50 (concentration inducing 50% cytotoxicity) of Py2Se2 in A549 and human normal lung fibroblast (WI38) cells was ∼8.5 μM and ∼5.5 μM, respectively, indicating that Py2Se2 did not exhibit selective toxicity towards cancer cells. Cell free studies indicated that Py2Se2 acted as a substrate of thioredoxin reductase (TrxR) and accordingly it was proposed that TrxR mediated reduction of Py2Se2 within cells might be generating intermediates leading to a reductive environment. Despite generating a reducing environment, Py2Se2 caused significant DNA damage, G1 phase arrest and apoptosis. The mechanistic investigation revealed that Py2Se2 induced G1 arrest was mediated through up-regulation of p21 transcript in a p53 independent manner. Further, the apoptotic effect of Py2Se2 was associated with the increase in the levels of unfolded protein response markers like BiP and CHOP, mitochondrial permeability (JC1) and apoptotic markers such as cleaved caspase-3 and poly (ADP-ribose) polymerase. Finally, pre-treatment with N-acetylcysteine (a stimulator of GSH biosynthesis) or L-buthionine sulfoximine (an inhibitor of GSH biosynthesis) increased and decreased the Py2Se2 mediated apoptosis, respectively. This confirmed that the cytotoxicity of Py2Se2 in A549 cells was triggered through reductive stress.


 Please wait while we load your content...
Please wait while we load your content...