Selenium deficiency exacerbates LPS-induced necroptosis by regulating miR-16-5p targeting PI3K in chicken tracheal tissue
Abstract
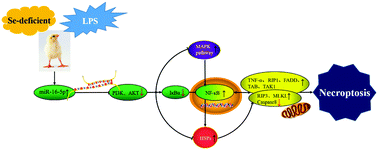
Multiple tissue necrosis is one of the morphological features of selenium deficiency-mediated injury. MicroRNA (miRNA) participates in the occurrence and development of necroptosis by regulating target genes. Necroptosis is a programmed form of necrosis, and it is closely related to lipopolysaccharide (LPS)-induced injury. Our aim was to investigate whether Se deficiency can promote tracheal injury caused by LPS through miRNA-induced necroptosis. By establishing models of tracheal injury in Se-deficient chickens, we verified the targeting relationship between chicken-derived miR-16-5p and PI3K through bioinformatics, qRT-PCR and WB analyses, and we measured the changes in the expression of genes related to the PI3K/AKT pathway, RIP3/MLKL pathway and MAPK pathway and of heat shock proteins. Under the condition of Se deficiency, the following results were observed: PI3K/AKT expression decreased with the upregulation of miR-16-5p, the expression of necroptosis-related factors (TNF-α, RIP1, FADD, RIP3 and MLKL) increased, and the expression of Caspase 8 significantly decreased (p < 0.05). Light microscopy observations indicated that cell necrosis was the main pathological change due to Se deficiency injury in the tracheal epithelium. The MAPK pathway was activated, and HSP expression was upregulated, indicating that the MAPK pathway and HSPs are both involved in Se deficiency-mediated necroptosis. In addition, Se deficiency promoted the expression of necroptosis-related genes in LPS-treated chickens (p < 0.05), and the pathological changes of cell necrosis were more obvious. In conclusion, we demonstrated that Se deficiency regulates the miR-16-5p-PI3K/AKT pathway and exacerbates LPS-induced necroptosis in chicken tracheal epithelial cells by activating necroptosis-related genes.


 Please wait while we load your content...
Please wait while we load your content...