Bis(ethylmaltolato)oxidovanadium(iv) inhibited the pathogenesis of Alzheimer's disease in triple transgenic model mice†
Abstract
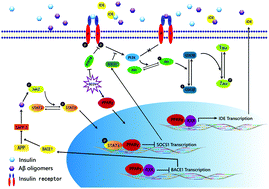
Vanadium compounds have been reported to mimic the anti-diabetes effects of insulin on rodent models, but their effects on Alzheimer's disease (AD) have rarely been explored. In this paper, 9-month-old triple transgenic AD model mice (3×Tg-AD) received bis(ethylmaltolato)oxidovanadium(IV) (BEOV) at doses of 0.2 mmol L−1 (68.4 μg mL−1) and 1.0 mmol L−1 (342 μg mL−1) for 3 months. BEOV at both doses was found to improve contextual memory and spatial learning in AD mice. It also improved glucose metabolism and protected neuronal synapses in the AD brain, as evidenced respectively by 18F-labeled fluoro-deoxyglucose positron emission tomography (18F-FDG-PET) scanning and by transmission electron microscopy. Inhibitory effects of BEOV on β-amyloid (Aβ) plaques and neuronal impairment in the cortex and hippocampus of fluorescent AD mice were visualized three-dimensionally by applying optical clearing technology to brain slices before confocal laser scanning microscopy. Western blot analysis semi-quantitatively revealed the altered levels of Aβ42 in the brains of wildtype, AD, and AD treated with 0.2 and 1.0 mmol L−1 BEOV mice (70.3%, 100%, 83.2% and 56.8% in the hippocampus; 82.4%, 100%, 66.9% and 42% in the cortex, respectively). The mechanism study showed that BEOV increased the expression of peroxisome proliferator-activated receptor γ (PPARγ) (140%, 100%, 142% and 160% in the hippocampus; 167%, 100%, 124% and 133% in the cortex) to inactivate the JAK2/STAT3/SOCS-1 pathway and to block the amyloidogenesis cascade, thus attenuating Aβ-induced insulin resistance in AD models. BEOV also reduced protein tyrosine phosphatase 1B (PTP1B) expression (74.8%, 100%, 76.5% and 53.8% in the hippocampus; 71.8%, 100%, 94.2% and 81.8% in cortex) to promote insulin sensitivity and to stimulate the PI3K/Akt/GSK3β pathway, subsequently reducing tau hyperphosphorylation (phosphorylated tau396 levels were 51.1%, 100%, 56.1% and 50.2% in the hippocampus; 22.2%, 100%, 36.1%, and 24% in the cortex). Our results suggested that BEOV reduced the pathological hallmarks of AD by targeting the pathways of PPARγ and PTP1B in 3×Tg AD mice.


 Please wait while we load your content...
Please wait while we load your content...