Insight into the antitumor actions of sterically hindered platinum(ii) complexes by a combination of STD NMR and LCMS techniques†
Abstract
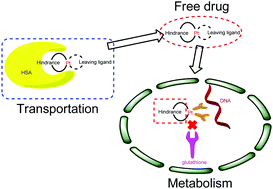
Sterically hindered platinum(II) complexes have shown great advantages in overcoming platinum drug resistance. In this study, the antitumor actions of sterically hindered platinum(II) complex 1 (cis-dichloro[(1R,2R)-N1-(2-fluorobenzyl)-1,2-diaminocyclohexane-N,N′]platinum(II), C13H19FPtCl2) were investigated by using saturation transfer difference nuclear magnetic resonance (STD NMR) and liquid chromatography–mass spectrometry (LCMS) techniques. STD NMR was applied to study the HSA (human serum albumin) binding properties, while the interactions between guanosine 5′-monophosphate (5′-GMP) and complex 1 were studied by LCMS. For HSA binding experiments, strong STD signals were observed for protons of sterically hindered parts of carrier ligands, indicating that the sterically hindered moieties of the carrier ligand could be situated inside the binding pocket of HSA. A 19F NMR experiment indicated that complex 1 could interact with HSA. Furthermore, the binding modes of complex 1 with guanosine 5′-monophosphate (5′-GMP) were studied in the absence and presence of glutathione by LCMS. According to the HPLC profiles, a mono-functional binding mode was observed for complex 1 both in the presence and in the absence of glutathione, while a bi-adduct was observed for Pt(DACH)Cl2, which may be one of the reasons for their different biological activities. Hence, this study demonstrated that the NMR method combined with the LCMS technique could provide valuable information to understand the transport and the underlying anticancer mechanisms of the platinum(II) complex at the molecular level. Moreover, the results reported here can help to reveal the binding mechanisms of the sterically hindered platinum(II) compounds with biomolecules, which may shed light on the design of novel platinum(II) anticancer agents with suitable sterically hindered groups.


 Please wait while we load your content...
Please wait while we load your content...