Melatonin triggers the anticancer potential of phenylarsine oxide via induction of apoptosis through ROS generation and JNK activation†
Abstract
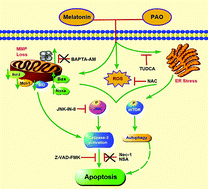
Melatonin, a safe endogenous hormone and a natural supplement, has recently been recognized to have antiproliferative effects and the ability to sensitize cells to other anticancer therapies. Phenylarsine oxide (PAO) has anticancer potential but it is considered as a toxic agent. In this study we combined melatonin to reduce the toxicity while securing the anti-cancer effects of PAO. Cell viability was determined by MTT assay, whereas cytotoxic assays were performed using an LDH cytotoxicity assay kit. Cell cycle analysis, Annexin V/PI staining, the mitochondrial membrane potential (MMP), mitochondrial calcium and reactive oxygen species (ROS) generation were analyzed using flow cytometry. Sytox stained cells were visualized by fluorescence microscopy and the expression of proteins was detected by western blotting. Melatonin increased the anticancer potential of PAO by decreasing the cell viability and increasing LDH release in various cancer cells. The mode of cell death was determined to be typical apoptosis, as evidenced by Annexin V/PI-stained cells, PARP cleavage, and caspase-3 activation, and with significant modulations in the expression of proapoptotic, antiapoptotic and cell cycle-related proteins. ROS generation played a critical role in induction of cell death by this combined treatment, which is validated by reversal of cytotoxicity upon cotreatment with NAC. Furthermore, the activation of MAPKs, especially JNK, contributed to the induction of cell death, accompanied by endoplasmic reticulum stress and autophagy, affirmed by the abrogation of cytotoxicity after JNK-IN-8 and TUDCA application. Melatonin showed promising potential as a chemotherapeutic agent in combination with PAO to achieve a better anticancer response.


 Please wait while we load your content...
Please wait while we load your content...