Serial in-solution digestion protocol for mass spectrometry-based glycomics and proteomics analysis†
Abstract
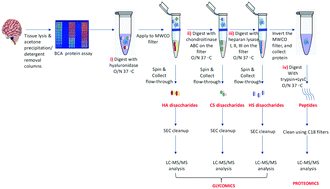
Advancement in mass spectrometry has revolutionized the field of proteomics. However, there remains a gap in the analysis of protein post-translational modifications (PTMs), particularly for glycosylation. Glycosylation, the most common form of PTM, is involved in most biological processes; thus, analysis of glycans along with proteins is crucial to answering important biologically relevant questions. Of particular interest is the brain extracellular matrix (ECM), which has been called the “final Frontier” in neuroscience, which consists of highly glycosylated proteins. Among these, proteoglycans (PGs) contain large glycan structures called glycosaminoglycans (GAGs) that form crucial ECM components, including perineuronal nets (PNNs), shown to be altered in neuropsychiatric diseases. Thus, there is a growing need for high-throughput methods that combine GAG (glycomics) and PGs (proteomics) analysis to unravel the complete biological picture. The protocol presented here integrates glycomics and proteomics to analyze multiple classes of biomolecules. We use a filter-aided sample preparation (FASP) type serial in-solution digestion of GAG classes, including hyaluronan (HA), chondroitin sulfate (CS), and heparan sulfate (HS), followed by peptides. The GAGs and peptides are then cleaned and analyzed using liquid chromatography-tandem mass spectrometry (LC-MS/MS). This protocol is an efficient and economical way of processing tissue or cell lysates to isolate various GAG classes and peptides from the same sample. The method is more efficient (single-pot) than available parallel (multi-pot) release methods, and removal of GAGs facilitates the identification of the proteins with higher peptide-coverage than using conventional-proteomics. Overall, we demonstrate a high-throughput & efficient protocol for mass spectrometry-based glycomic and proteomic analysis (data are available via ProteomeXchange with identifier PXD017513).
- This article is part of the themed collection: Glycomics & Glycoproteomics: From Analytics to Function


 Please wait while we load your content...
Please wait while we load your content...