Rapid and sensitive glycan targeting by lectin-SERS assay†
Abstract
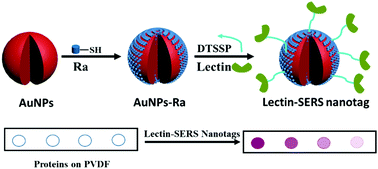
Glycosylation is an important part of cell signalling that is implicated in many disease states in which glycans play an essential role. Therefore rapid and sensitive differentiation of glycans on proteins is highly desirable. Current technologies for glycan structural analysis normally involve the isolation of glycans from proteins, or enrichment of glycopeptides, and detection by mass spectrometry, which requires relatively large amounts of sample and is not able to be used by non-specialist laboratories. Herein we present a simple and new strategy for targeting the glycans on a protein (with IgG as a model glycoprotein) using surface-enhanced Raman scattering (SERS) coupled to glycan-binding WGA (wheat germ agglutinin) lectin, in a lectin-SERS assay. With one drop (1 μL) of glycoprotein solution, our lectin-SERS assay can detect as low as 10 ng IgG within two hours with high glycan specificity. We extend our technique to examine the surface glycan profiles on two human colorectal cancer cell lines, which show different and unique glycan signatures specific to the target cell lines. Thus, we believe that this method could be potentially used for the real-time and in situ monitoring of glycans on the surface of cells or tissue or in body fluids, and is thus a powerful tool for glycomics research.
- This article is part of the themed collection: Glycomics & Glycoproteomics: From Analytics to Function


 Please wait while we load your content...
Please wait while we load your content...