Mycobacterium bovis BCG infection alters the macrophage N-glycome†
Abstract
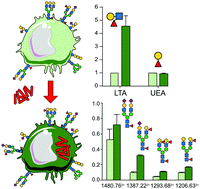
Macrophage glycosylation is essential to initiate the host-immune defense but may also be targeted by pathogens to promote infection. Indeed, the alteration of the cell-surface glycosylation status may affect the binding of lectins involved in cell activation and adhesion. Herein, we demonstrate that infection by M. bovis BCG induces the remodeling of the N-glycomes of both human primary blood monocyte-derived macrophages (MDM) and macrophage-cell line THP1. MALDI-MS based N-glycomic analysis established that mycobacterial infection induced increased synthesis of biantennary and multifucosylated complex type N-glycans. In contrast, infection of macrophages by M. bovis BCG did not modify the glycosphingolipids composition of macrophages. Further nano-LC-MSn glycotope-centric analysis of total N-glycans demonstrated that the increased fucosylation was due to an increased expression of the Lex (Galβ1-4[Fucα1-3]GlcNAc) epitope, also known as stage-specific embryonic antigen-1. Modification of the surface expression of Lex was further confirmed in both MDM and THP-1 cells by FACS analysis using an α1,3-linked fucose specific lectin. Activation with the mycobacterial lipopeptide Pam3Lp19, an agonist of toll-like receptor 2, did not modify the overall fucosylation pattern, which suggests that the infection process is required to modify surface glycosylation. These results pave the way toward the understanding of infection-triggered cell-surface remodeling of macrophages.
- This article is part of the themed collection: Glycomics & Glycoproteomics: From Analytics to Function


 Please wait while we load your content...
Please wait while we load your content...