Harnessing the self-assembly of peptides for the targeted delivery of anti-cancer agents
Abstract
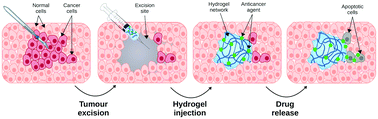
A significant challenge to current cancer drug treatment is mode of delivery, both in terms of efficacy and off-target toxicity to healthy tissues. To overcome this, drug localisation using a range of biocompatible carriers is currently in use or under investigation. One class of these biomaterial carriers that offers a unique prospect for use as drug delivery vectors to tumour sites is hydrogels formed by small molecules. In particular, tissue mimetic self-assembling molecular hydrogels can function either as injectable precursors that gelate in response to tumour-specific markers, or as implants in conjunction with surgical resection or tumour debulking. Their inherent biocompatibility, tuneable properties, and capacity to flow and gelate in situ allow them to effectively transport, hold and release therapeutic molecules in a spatially and temporally controlled manner. This has been shown in a number of in vitro and in vivo studies, where they improve anti-cancer efficacy while reducing non-specific toxicity. However, further investigation is required to optimise these systems toward both the drug and the target tissue, to provide sophisticated temporal control over the drug presentation, and to determine the most effective drug–material combinations for specific cancer types and locations.


 Please wait while we load your content...
Please wait while we load your content...