Enhanced and unconventional responses in chemiresistive sensing devices for nitrogen dioxide and ammonia from carboxylated alkylthiophene polymers†
Abstract
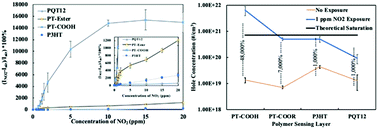
A carboxylated thiophene polymer-based chemiresistive device in a field-effect transistor (FET) configuration with unusual and enhanced responses to the widespread pollutants nitrogen dioxide (NO2) and ammonia (NH3) is described. The device based on a polymeric thiophene carboxylic acid showed a dramatic and superlinear increase in drain current (ID) of over 15 000% to a ramped exposure to 10 ppm NO2 over several minutes, while its ethyl ester counterpart had significantly lower response. Devices incorporating either an ester or carboxylic acid displayed comparable and previously unreported increases in ID from 10 ppm ramped NH3 exposure of 200–300%. Conventional poly(alkylthiophenes) showed the expected current decreases from similar NH3 exposures. Using threshold voltage shifts in silicon transistors coupled to our recently reported remote gate (RG) platform with thiophene polymer coatings, we determined that two differing response mechanisms are associated with the two gas exposures. By calculating the charge density induced in the polymers by NO2 exposure using the silicon transistor voltage shifts, we conclude that proton conduction contributes significantly to the high sensitivity of the carboxylic acid to NO2, in addition to doping that was observed for all four polymers. Furthermore, hydrogen bonding moieties of the carboxylic acid and ester may be able to physisorb NH3 and thus alter the charge distribution, rearrange polymer chains, and/or create a proton transfer network leading to the ID increase that is the opposite of the response obtained from non-carboxylated thiophene polymers.


 Please wait while we load your content...
Please wait while we load your content...