Bulk ion-clustering and surface ion-layering effects on work function of self-compensated charged-doped polymer semiconductors†
Abstract
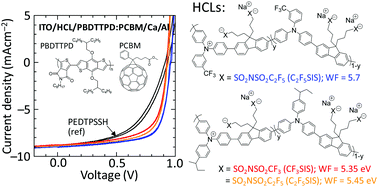
Hole- and electron-doped polyelectrolytes are of much current interest because they afford workfunction-tuneable charge injection layers that are immune to ‘dopant’ migration. However the role of the covalently tethered ion beyond providing solubility in these systems have not been well understood. Here using two well-known fluorene-alt-triarylamine model semiconductor cores (TFB and mTFF), we show, using variable-angle X-ray and ultraviolet photoemission spectroscopies, that the larger tethered anions—trifluoromethanesulfonylimidosulfonyl and pentafluoroethanesulfonylimidosulfonyl—give stronger surface ionic layering than sulfonate. This contributes to increasing both the HOMO ionization energy of the undoped polymer, and the vacuum work function (WF) of the hole-doped polymer, by about 0.2 eV, which provides the desired ‘fine tuning’ of these parameters. In addition, molecular dynamic simulations of the ion cluster structure in the bulk show that the larger anions produce a larger average distance to the holes. This raises the electrochemical energy of the holes, advantageously increasing their inherent WF by another 0.1 eV or so. Device electroabsorption spectroscopy shows that the increased WF persists into devices, resulting in better hole injection efficiency into large-ionization-energy organic semiconductor layers, and better hole collection efficiency from organic solar cells. Finally, the larger tethered anions also impart better solubility to the hole-doped form in fluorinated alcohols, acetonitrile and propylene carbonate, overcoming a key bottleneck for the deployment of these materials in practical applications.


 Please wait while we load your content...
Please wait while we load your content...