Unveiling the underlying mechanism of record-high efficiency organic near-infrared photodetector harnessing a single-component photoactive layer†
Abstract
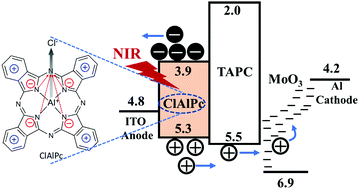
This paper demonstrates high-efficiency organic near-infrared (NIR) photodetection activity by increasing the symmetry of photoinduced charge density based on planar cyclic donor–acceptor structures. The record-high photosensing capability (gain: 3 × 105 and EQE: 43% at 3 V) is realized under NIR 780 nm illumination upon using the single-component active layer chloroaluminum phthalocyanine (ClAlPc) with planar cyclic intramolecular donor–acceptor structures to create four-fold symmetry of photoinduced charge density. Our in situ measurement using bias-dependent magneto-photocurrent reveals that increasing the symmetry of photoinduced charge density through planar cyclic intramolecular donor–acceptor structures can significantly decrease the electron–hole binding energies, allowing the direct generation of free photogenerated carriers without any heterojunction structures. Particularly, with the four-fold symmetry of photoinduced charge density, the dissociation of electron–hole pairs can be completed at an extremely low critical bias (24.8 mV) in the ClAlPc molecules. Such low critical bias provides the underlying mechanism to directly generate free carriers in the single-component active layer under NIR illumination. Clearly, by using planar cyclic intramolecular donor–acceptor structures, symmetrically arranging photoinduced charge density presents a fundamental strategy to develop high-efficiency NIR sensing capabilities with single-component active layer design in organic molecules.


 Please wait while we load your content...
Please wait while we load your content...