A zinc bromine “supercapattery” system combining triple functions of capacitive, pseudocapacitive and battery-type charge storage†
Abstract
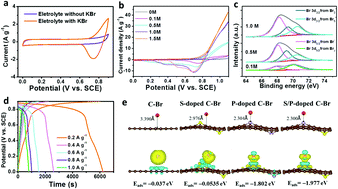
Advanced electrochemical energy storage devices are in high demand to meet the need for both short-term high power pulse and long-term durable energy outputs. To achieve this goal, a majority of studies have focused on optimization of solid electrode materials. However, the issue of slow diffusion of ions in electrode materials causes unsatisfactory power density in the energy storage devices. In this work, soluble dual-redox additives in an aqueous electrolyte were employed to combine with a S/P co-doped carbon based cathode and a carbon cloth (CC) based anode to construct an aqueous zinc bromine (Zn–Br2) “supercapattery”. The as-fabricated supercapattery possessed electrical-double-layer type, pseudocapacitive, and battery-type charge storage in both cathode and anode and successfully united capacitive character and battery-like character in one device. The benefits of the triple functions of charge storage resulted in an extremely high energy density of 270 W h kg−1 and a maximum power density of 9300 W kg−1. The capacitive and pseudocapacitive charge storage processes in the Zn–Br2 supercapattery are able to provide peak power output. Meanwhile, the diffusion-controlled battery-type redox reactions can meet the requirement for high energy. This work provides a new pathway towards development of electrochemical energy storage devices with both battery-level energy density and capacitor-level power density.


 Please wait while we load your content...
Please wait while we load your content...