Automated microfluidic screening of ligand interactions during the synthesis of cesium lead bromide nanocrystals†
Abstract
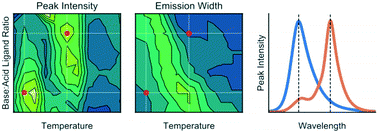
Varying the nature of the molecular surface ligands used to synthesize and stabilize lead halide perovskite nanocrystals provides a facile way of tuning and optimizing particle properties. However, the inherently complex reaction parameter space associated with perovskite nanocrystal synthesis complicates this, necessitating long-term and laborious experimentation to optimize conditions for a specific ligand set, and to compare nanoparticle properties across different ligand sets. In the current work, we present an automated microfluidic-based strategy for multidimensional parameter screening, seeking to maximize luminescence intensities and minimize emission bandwidths from quantum confined nanostructures, by monitoring variations in photoluminescence as a function of reaction temperature, base-to-acid ligand ratio, and ligand alkyl chain structure (linear versus branched). For a linear ligand pair (octylamine and octanoic acid), we find two reaction parameter sets that yield bright and narrow emission from nanosheets emitting at 460 nm (synthesized at 100 °C with a low base : acid ratio, FWHM = 25 nm) and nanowires emitting at 497 nm (synthesized at 140 °C with a high base : acid ratio, FWHM = 17 nm). Introducing branched ligands (2-ethylhexylamine and/or 2-ethylhexanoic acid) induces a red shift in emission and shows that bright and narrow emission can only be obtained from weakly quantum-confined nanostructures, within the reaction conditions studied. This work represents the first use of a rapid automated microfluidic system for the screening of ligand interactions during the synthesis lead halide perovskite nanocrystals.
- This article is part of the themed collection: 2021 MSDE Symposium Collection


 Please wait while we load your content...
Please wait while we load your content...