Connecting molecular mechanisms of biofilm formation and functional biomaterials
Abstract
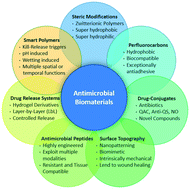
Biofilms are arguably the root cause of most human infections, imposing a high level of public health concern. Biofilm growth is a lifestyle characterized by co-aggregation of organisms and secretion of an extracellular matrix (ECM) that facilitates attachment to a surface. They are responsible for up to 80% of all healthcare-acquired infections (National Institutes of Health) with the majority being diagnosed as device associated. They are multi-compositional structures that are influenced by surrounding biological, mechanical and chemical variation in their microenvironment. Within the confines of biofilms, microorganisms are capable of promoting increased rates of antimicrobial resistance. When observing biofilm physiology in the presence of a biomaterial, the attachment process and intermolecular activity, contribute to significant genotypic responses. When utilized as part of medical devices, antimicrobial biomaterials seek to prevent biofilm infections, however, the complexity of biofilms make it difficult to extract useful results necessary to improve material properties without consideration of bacterial molecular processes. This interdisciplinary article emphasizes molecular mechanisms as a pertinent consideration in evaluating biofilm growth on biomaterials. It also advocates for standardizing in vitro biofilm models as opposed to animal models. Such models include microfluidic systems, that can be engineered to scale and mimic infection-relevant microenvironments. The connection of advances in antibiofilm surface modifications and high-throughput sequencing make way for new and important biomaterial assessments. We endeavor to establish a gold standard model for in vitro biomaterials testing that incorporates the complexity of healthcare-associated infections in order to guide medical device design and improve disease outcomes.


 Please wait while we load your content...
Please wait while we load your content...