Magnetism modulation of Co3S4 towards the efficient hydrogen evolution reaction
Abstract
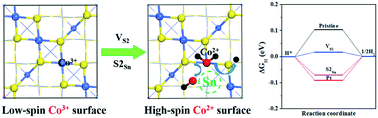
Transition metal sulfides have been widely used as highly-efficient and low-cost electrocatalysts for water splitting. We performed systematic first-principles studies for the structural stability, hydrogen evolution reaction activity and water dissociation on pristine and decorated Co3S4(001) surfaces. We found that the creation of one kind of S-vacancy and its substitution with Sn can be two effective ways to enhance the electrocatalytic performance of the Co3S4(001) surface, as well as to drop the energy barrier for water dissociation. The mechanism behind this is the appearance of high spin Co2+ atoms adjacent to a vacancy or dopant, which induce magnetism to sulfur atoms and activate more reaction sites. Our results gain insights into modulating and designing new transition metal sulfides for their applications in electrocatalysts.


 Please wait while we load your content...
Please wait while we load your content...