Selective chemical adsorption of Cd(ii) on silica covalently decorated with a β-ketoenol-thiophene-furan receptor
Abstract
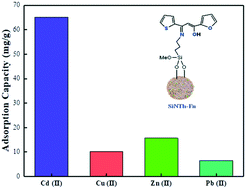
Nowadays, porous hybrid materials are considered as potential reservoirs of metallic species in environmental clean-up technologies. Herein, a new environment-friendly surface with thiophene-furan-β-ketoenol grafted on a colloidal silica surface has been synthesized with the purpose of using it as an effective and selective adsorbent for Cd(II) from aquatic environments. Elemental analysis, Fourier-transform infrared spectroscopy, scanning electronic microscopy, thermogravimetric analysis and Brunauer–Emmett–Teller analysis confirm the successful surface incorporation of the β-ketoenol bis-heterocycle receptor. The novel inorganic–organic hybrid material was applied for Cd(II), Cu(II), Zn(II) and Pb(II) adsorption. Cd(II) adsorption reaches 84.45 mg g−1 within only 30 min at pH 6, and the adsorption was more appropriate with the 2nd order kinetic model (R2 ≥ 0.997) and Langmuir model isotherm where the adsorption process is coherent with a monolayer adsorption reaction. The ΔH°, ΔS° and ΔG° parameters reflect spontaneous, and endothermic adsorption and that the process increases the randomness. The new material demonstrates its efficiency and selectivity towards Cd(II) and promises good reusability for at least five elimination cycles. The new material can be considered as a reliable cleaner of Cd(II) from aquatic environments.
- This article is part of the themed collection: Molecular systems for sensing


 Please wait while we load your content...
Please wait while we load your content...