Arylsulfonyl histamine derivatives as powerful and selective α-glucosidase inhibitors†
Abstract
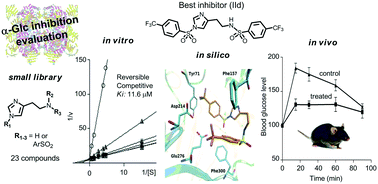
A series of simple N-arylbenzenesulfonyl histamine derivatives were prepared and screened against α-glucosidase. Inhibition was in the micromolar range for several Nα,Nτ-di-arylsulfonyl compounds, with Nα,Nτ-di-4-trifluorobenzenesulfonyl histamine (IId) being the best inhibitor. Compound IId is a reversible and competitive α-glucosidase inhibitor, and presented good selectivity with respect to other target enzymes, including β-glucosidase and α-amylase, and interesting predicted physicochemical properties. Docking studies have been run to postulate ligand–enzyme interactions to account for the experimental results. In vivo, compound IId produced a similar hypoglycemic effect to acarbose with half of its dose.
- This article is part of the themed collection: Celebrating Latin American Talent in Chemistry


 Please wait while we load your content...
Please wait while we load your content...