Aromadendrin: a dual amyloid promoter to accelerate fibrillization and reduce cytotoxicity of both amyloid-β and hIAPP
Abstract
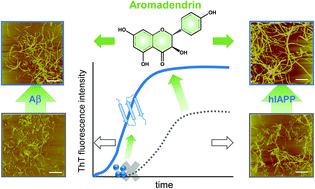
Abnormal aggregation of misfolded amyloid proteins into amyloid fibrils is associated with many neurodegenerative diseases, including Alzheimer's disease (AD) and Type II diabetes (T2D). In principle, any strategy to alter the amyloid aggregation process is considered as a potential therapeutic treatment for these diseases. Significant efforts and progress have been made to develop amyloid inhibitors that can slow down and prevent the amyloid aggregation process. However, much less research has been reported to discover general amyloid promoters to accelerate amyloid progress and remodel toxic amyloids. Here, for the first time we report a repurposing of the drug of aromadendrin as a dual amyloid promoter to be effective in accelerating amyloid aggregation/fibrillization and reducing neuroblastoma/insulinoma toxicity of both Aβ42 (associated with AD) and hIAPP37 (associated with T2D). ThT, AFM, and CD results showed that addition of aromadendrin to amyloid solutions with 1 to 5 molar ratios caused significant acceleration in Aβ42 fibrillization by 86–114% and hIAPP fibrillization by 20–68% as evidenced by shortening or bypassing of the lag phase, promoting the growth phase, and rapidly converting the amyloid species towards the higher ordered β-structure-rich aggregates. Seeding experiments further revealed that aromadendrin is more effective in accelerating the aggregation and structural conversion of the amyloid species at the lag and early growth phases, but unable to escalate the fibrillization of higher order protofibrils. Moreover, MTT and LDH cell assays showed that aromadendrin-treated cell samples enabled the rescue of cells from both Aβ- and hIAPP-induced toxicity by increasing cell viability by 12–15% (Aβ) and 10–49% (hIAPP) and reducing cell apoptosis by 45–67% (Aβ) and 10–30% (hIAPP). LUV assays explained the protective role of aromadendrin in cell toxicity due to the suppression of toxic oligomer formation and thus the decrease of membrane leakage. This work offers a new strategy for repurposing aromadendrin as an amyloid promoter, not as an amyloid inhibitor, to accelerate amyloid formation and remedy the amyloid-mediated toxicity for both Aβ and hIAPP, and the sequence-independent promotion effect of aromadendrin could also be applied to other amyloid proteins.


 Please wait while we load your content...
Please wait while we load your content...