Origin of Sn(ii) oxidation in tin halide perovskites†
Abstract
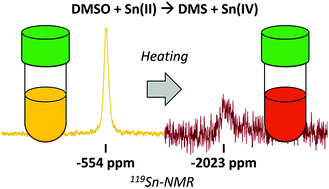
Tin-halide perovskites have great potential as photovoltaic materials, but their performance is hampered by undesirable oxidation of Sn(II) to Sn(IV). In this work, we use nuclear magnetic resonance spectroscopy (NMR) to identify and describe the origins of Sn(IV) in Sn-based perovskites, mainly focusing on direct measurements of Sn oxidation states with 119Sn-NMR in solid-state and solution. We find that dimethylsulfoxide (DMSO), a typical solvent for Sn-based perovskites, oxidizes Sn(II) in acidic conditions under temperatures used for film annealing. We propose a redox reaction between DMSO and Sn(II), catalyzed by hydroiodic acid, with iododimethylsulfonium iodide intermediate. We find that lower temperatures and less acidic conditions abate this reaction, and we assess a range of compositions and solution components for this instability. These results suggest the need for strategies to prevent this reaction and shed light on other solution instabilities beyond Sn(IV) that must be mitigated to achieve high-performance lead-free perovskites.
- This article is part of the themed collections: Advances in Materials Characterisation, Perovskites and International Open Access Week 2020


 Please wait while we load your content...
Please wait while we load your content...