Controlling interface properties for enhanced photocatalytic performance: a case-study of CuO/TiO2 nanobelts†
Abstract
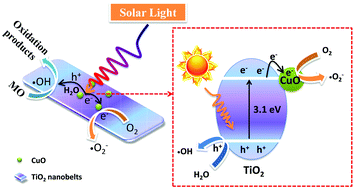
TiO2 nanobelts with CuO modification were designed and fabricated via a facile strategy. The photocatalytic degradation of methyl orange (MO) is selected as a model reaction to investigate the photocatalytic performance of all as-prepared CuO/TiO2 nanobelts under full-spectrum light (300–2500 nm) irradiation. The optimized mass fraction of CuO is 0.1% for CuO/TiO2 composite nanobelts, namely CT-0.1 sample, which can almost completely degrade MO pollutant in 50 min. The as-obtained CuO/TiO2 composites are systematically investigated by a variety of physical and chemical characterizations. Therein, it can be obtained that superoxide radicals (˙O2−) and hydroxyl radicals (˙OH) are the main active species in this photocatalytic system. The photoelectrochemical measurement clearly demonstrates that the enhanced photocatalytic activity can be attributed to the efficient separation and transfer of photo-generated electron–hole pairs, and lower overpotential for CuO/TiO2 nanobelts. This work provides a prototype to study the photocatalytic oxidation process, which contributes to the design and construction of highly-efficient composite photocatalysts.


 Please wait while we load your content...
Please wait while we load your content...