Probing the charged nature and ion-exclusion mechanism of fluorine-enriched non-ionogenic polyamide derived thin film composite nanofiltration membranes†
Abstract
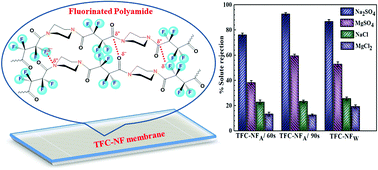
A thin film composite (TFC) nanofiltration (NF) membrane comprising a novel fluorine-enriched polyamide (FPA) as a skin layer polymer has been developed by in situ interfacial polycondensation between a cyclo-aliphatic diamine monomer, piperazine, and an acyclic fluorodiacyl chloride monomer, tetrafluorosuccinyl chloride. Instrumental investigations carried out by FTIR and XPS showed that the structure of FPA is devoid of intra- as well as inter-molecular H-bonds, which seems to be a rare finding in NF type membranes with decent performances. Stereoelectronic effects of the FPA structure, owing to the configurational mobility of polymer chains, electron-withdrawing ability of fluorine and lack of cohesive non-covalent type interactions, strongly influenced the surface morphology, topography and hydrophilicity of the TFC-NF membranes. The active skin layer of the membranes exhibited uniform charge-atmosphere in spite of not having any fixed charged groups in the polymer structure constituting it. Zeta potential measurement of the membranes revealed low values (−4.4 to −7 mV), indicating their low surface charge. However, a high Donnan-exclusion was observed towards a solute with multivalent anion (Na2SO4, solute rejection: 87–93%) as opposed to that with monovalent anion (NaCl, solute rejection: 23–26%), with a solvent flux as high as 54 L m−2 h−1 at 15 bar transmembrane pressure. The current class of TFC-NF membranes with unique physicochemical attributes thus provides a promising scope for a variety of nanofiltration applications.


 Please wait while we load your content...
Please wait while we load your content...