Advanced 96-microtiter plate based bioelectrochemical platform reveals molecular short cut of electron flow in cytochrome P450 enzyme†
Abstract
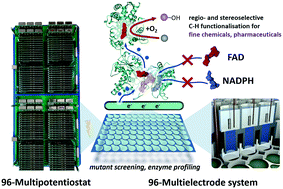
In bioelectrocatalysis, immobilised redox enzymes are activated in a bioelectronic interface without redox equivalents such as NADPH, thus enabling heterogeneous flow chemistry. The functional contact between enzyme and electrode requires a high degree of optimisation regarding choice of electrode material, electrode pre-treatment, enzyme immobilisation and reaction conditions. So far, however, there are no systems that can easily enable an optimisation procedure at a higher throughput. Here, we present an advanced platform with a vertical divided cell architecture in conjunction with a developed 96-multipotentiostat to be able to drive redox enzymes in 96 well microtiter plate based multielectrode arrays. This platform controls 96 independent three-electrode setups with arbitrary working electrode materials. We demonstrate its applicability in a mutation study of cytochrome P450 BM3 using indium tin oxide as electrode material and the 7-ethoxycoumarin product quantification assay. We show that the bioelectrocatalytic activity of P450 BM3 can be amplified when the cofactor FAD is erased from the enzyme by a single point mutation, so that FMN becomes the first electron entry point. Bioelectrocatalysis thus offers an approach to enzyme simplification as a remedy for the inherent instability of self-sufficient cytochrome P450 enzymes. In addition, we examined native and artificial enzyme activation with respect to ionic strength and buffer composition. The optimal conditions of the activation types differ substantially from each other and exhibit a new molecular facet in enzyme characteristics. In a proof-of-principle we demonstrate that the platform is also compatible with raw cell extracts, thus opening the door for random mutagenesis screenings.


 Please wait while we load your content...
Please wait while we load your content...