Nitric acid effect in inductively coupled plasma mass spectrometry: new insights on possible causes and correction†
Abstract
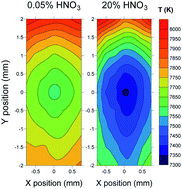
Fundamental aspects related to the analysis of nitric acid solutions by inductively coupled plasma mass spectrometry have been critically revisited in order to provide new insights into the possible sources of non-spectroscopic matrix effects and their correction by internal standardization. For elements having first ionization potential (IP) lower than 7.5 eV, the increase in the signal due to nitric acid was ascribed to a delay in the ionization process, which resulted in a reduction of the ion lateral diffusion in the plasma. In contrast, for non-easily ionisable elements, signal depression was correlated with their IP value and attributed to the cooling effect due to the energy required for nitric acid dissociation. Moreover, the atomic mass did not show any significant effect, thus precluding the application of the widely accepted criterion of choice of internal standards based on matching of masses. The extent and sign of the acid effect also depended on the applied operating conditions. For elements with low IP (e.g. Co, Cs, In, Li, Pb, Y), nitric acid caused signal enhancement at high RF power and nebulizer gas flow rate values (i.e., plasma robust conditions) and signal depression under non-robust conditions. For elements with high IP (e.g. As, Be, Cd and Sb), the decreasing acid effect became stronger with increasing the nebulizer gas flow rate and decreasing the RF power. The acid effect was also influenced by the ion lens voltage, that significantly interacted with the RF power effect. Finally, a procedure for the selection of optimal internal standards by principal component analysis has been successfully applied for the correction of the nitric acid effect.
- This article is part of the themed collection: Community Leaders: Gary Hieftje


 Please wait while we load your content...
Please wait while we load your content...