Evidence for photochemical synthesis of fluoromethane
Abstract
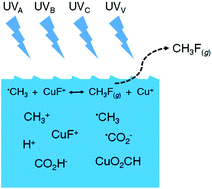
Photochemical generation of volatile fluoromethane in a dilute acetic acid medium is demonstrated. Radical halogen atom transfer (XAT) from an intermediate Cu–F transition metal fluorocomplex to abundant ˙CH3 generated by UV homolysis of acetic acid, rather than direct formation of free ˙F radicals, is hypothesized as being the synthetic route. The presence of Cu(II) is indispensable for CH3F formation. GC-MS identification of the gaseous products released from a photolysed 2% (v/v) solution of acetic acid containing 60 mg L−1 added Cu(II) as a sensitizer confirms synthesis of CH3F as the volatile fluorinated species, which is not detectable in the absence of Cu(II). Reaction efficiency remains unchanged whether the sample medium is exposed to V-UV radiation (185 nm) or germicidal UV-C radiation (primarily 254 nm and longer wavelengths) from the source. Experimental optimization of generation conditions (pH, irradiation time, UV wavelength, acetic acid concentration, metal ion sensitizer and concentration) was undertaken with a view to elucidation of mechanism as well as maximizing synthetic yield. Although the low yield (<1%) of CH3F currently precludes its efficacious use as a means of enhancing analyte introduction efficiency into atomic spectrochemical sources, this study demonstrates successful application of XAT to encompass PVG of the most difficult of the halogens to process in this manner and may provide insights into further studies to ultimately achieve higher yields.
- This article is part of the themed collection: Community Leaders: Gary Hieftje


 Please wait while we load your content...
Please wait while we load your content...