Rapid determination of Ba isotope compositions for barites using a H2O-extraction method and MC-ICP-MS†
Abstract
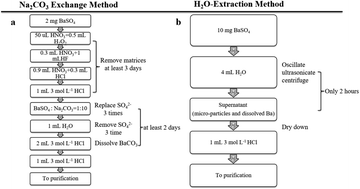
This study presents a rapid and simple method of high precision Ba isotope measurement for barite using H2O extraction. The main challenge for analyzing the Ba isotope composition of barite is how to digest barite into solution for chemical purification. We used H2O as a dispersant to rapidly extract ∼20–75 μg Ba as barite micro-particles (invisible to the naked eye) into suspension, and then these micro-particles were quickly dissolved in 3 mol L−1 HCl. The precision and accuracy of this method were rigorously evaluated by two ways. Firstly, we analyzed four synthetic barites made by mixing different proportions of pure barite and matrix rocks (black shale, carbonatite, and clastic rock) with high Ba content and different δ138/134Ba. Despite the variable proportions of barite in the synthetic standards (10%, 25%, 40%, and 70%), the average measured δ138/134Ba is 0.33 ± 0.03‰ (2SD, n = 8), well consistent with that of a pure barite standard (GBW07812, 0.33 ± 0.05‰, 2SD, n = 36). Secondly, we repeatedly analyzed five barite standards (NBS127, GBW07811, GBW07812, GBW07814, and GBW07816). The Ba isotope compositions of these barites determined by the H2O extraction method are well consistent with those previously measured by the Na2CO3 exchange method. The long-term external precision of δ138/134Ba is better than 0.05‰ (2SD). Compared with the Na2CO3 exchange method, the H2O-extraction method significantly simplifies the digestion procedure and improves the efficiency of barite Ba isotope analysis.


 Please wait while we load your content...
Please wait while we load your content...