An improved separation scheme for Sr through fluoride coprecipitation combined with a cation-exchange resin from geological samples with high Rb/Sr ratios for high-precision determination of Sr isotope ratios
Abstract
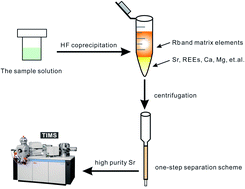
A cation-exchange resin is commonly used to separate Sr from geological and environmental samples. AG50 resin is the most traditional cation-exchange resin and has the advantages of good reusability and low cost. Although AG50 resin can meet the demands of most conventional geological samples, eliminating 87Rb isobaric interference from samples with high Rb/Sr ratios (>30) is difficult during one-step separation. Separating Sr twice using AG50 resin is the general approach to Rb removal. The two-step separation method is time consuming and leads to a low Sr yield. This study aimed to improve the sample throughput and overcome the problem of 87Rb isobaric interference from imperfect separation. A new separation method involving hydrofluoric acid (HF acid) coprecipitation combined with a cation-exchange resin (i.e. AG50 resin) for samples with high Rb/Sr ratios was developed. HF acid was used as a coprecipitator to remove most Rb (>90%) and recover Sr before performing AG50 column chemistry. The reliability of this method was examined using the rhyolite standard JR-2, a typical sample with a high Rb/Sr ratio (Rb/Sr = 37.36). Results in the JR-2 case showed that approximately 92% of Rb and most of the matrix elements (K, Ti, Fe, Al, Mn and Na) could be removed effectively. Moreover, Sr recovery exceeded 91% during HF acid coprecipitation. After the removal of Rb by HF acid coprecipitation, the Sr fraction with high purity was easily obtained through one-step separation using the AG50 resin column. However, we found that the removal rates of Rb and some matrix elements (such as Al and Na) were strongly affected by the composition of Al–Ca–Mg in samples. Our separation method is more suitable for samples with high Rb/Sr ratios and low Ca and Mg contents. A series of silicate rock reference materials were selected to evaluate the applicability of our method, and the Sr isotopic results were consistent with previously reported values. Overall, this rapid, simple and low-cost method shows great potential for the separation of Sr from geological samples with high Rb/Sr ratios, such as alkaline feldspar granite, alkaline granite and alkaline rhyolite.


 Please wait while we load your content...
Please wait while we load your content...