A heterogeneous and recoverable palladium catalyst to access the regioselective C–H alkenylation of quinoline N-oxides†
Abstract
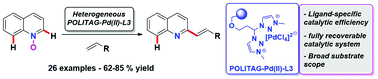
Herein, we disclose the first C-2-selective C–H alkenylation of quinoline N-oxides catalyzed using a heterogeneous palladium catalyst. The protocol does not require the use of an external oxidant and it is applicable to an ample substrate scope always showing excellent site selectivity. This process is made accessible by the use of a specific 1,2,3-triazolium-tagged heterogeneous polymeric catalytic system. The catalyst can be efficiently recovered and reused with no decrease of its catalytic performance and hot filtration and mercury poisoning tests suggest that its mechanism of action is operatively heterogeneous. In addition, mechanistic studies revealed that C–H activation reaction pathways are operative, setting the stage for the direct synthesis of 2-functionalized quinolines using N-oxide functionality as both a directing group and an oxidant.


 Please wait while we load your content...
Please wait while we load your content...