Supercritical CO2-induced alteration of a polymer–metal matrix and selective extraction of valuable metals from waste printed circuit boards
Abstract
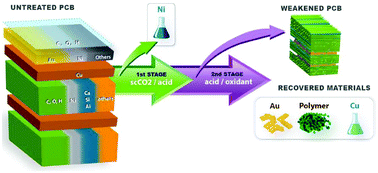
The rapid accumulation of waste electrical and electronic equipment (WEEE) is one of the biggest environmental concerns in modern societies, and this problem will be further accelerated in the future. The use of supercritical CO2 (scCO2) mixed with acids has been proposed as a greener solvent system compared to conventional cyanide and aqua regia solvents. However, the mechanisms of scCO2 for metal extraction from WEEE are still poorly understood. Thus, this study focused on the physical, structural, and chemical interactions between scCO2/acid solvents and complex layered components in waste printed circuit boards (WPCBs), a common type of WEEE. Our study showed that the use of scCO2-based pretreatment allows faster leaching of metals including copper (Cu) in the subsequent hydrometallurgical process using H2SO4 and H2O2, while allowing gold (Au) recovery as hydrometallurgically delaminated solids. This enhancement is due to the selective leaching of Ni and unique inner porous structures created by ScCO2/acid treatment via dissolving the calcium-silicate-bearing fiberglass within WPCBs. Thus, the scCO2-based pretreatment of WPCBs shows a multifaceted green chemistry potential relating to the reduction in solvent usage, and targeted recovery of Au prior to shredding or grinding that would reduce any loss or dilution of Au in the subsequent waste stream.


 Please wait while we load your content...
Please wait while we load your content...