Visible light-catalytic hydroxylation of aryl halides with water to phenols by carbon nitride and nickel complex cooperative catalysis†
Abstract
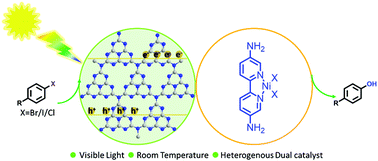
The efficient conversion of aryl halides to phenols under mild reaction conditions remains a great challenge. Here we demonstrate that a facile and efficient hydroxylation reaction of aryl halides with water could be accomplished through a merger of a heterogeneous organic semiconductor graphitic carbon nitride (g-C3N4) photocatalyst and a homogeneous nickel(II) bipyridine organometallic catalyst under visible-light irradiation at room temperature. The protocol is tolerant to a wide range of substituted aryl halides, resulting in various phenol products with excellent conversion and selectivity. It is worth noting that the OH group of the resulting phenols originating from water increased the environmental friendliness of the reaction. It is worth noting that among all the previously reported catalysts (even including thermal catalysts with harsh reaction conditions), our Nidabpy/g-C3N4 dual catalytic system demonstrated the highest catalytic activity reported in the shortest reaction time. This remarkable catalytic performance was attributed to the strong interaction between the Nidabpy complex and g-C3N4, which decreased the transfer distance of photogenerated electrons transferring to Ni(II), accelerating the catalytic cycle. Furthermore, g-C3N4 was easily recovered from the reaction mixture and reused five times, showing the good potential of green hydroxylation reactions for large-scale industrial application.


 Please wait while we load your content...
Please wait while we load your content...