A simple method for producing bio-based anode materials for lithium-ion batteries†
Abstract
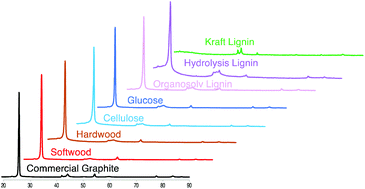
A simple and scalable method for producing graphite anode material for lithium-ion batteries is developed and demonstrated. A low-cost, earth abundant iron powder is used to catalyze the conversion of softwood, hardwood, cellulose, glucose, organosolv lignin, and hydrolysis lignin biomaterials to crystalline graphite at relatively low temperatures (<1200 °C). Biographite materials are characterized and compared based on graphite mass yield, graphite crystallite size, degree of graphitization, graphite uniformity, iron catalyst distribution, and graphite morphology. Particle size, heating method, and intermediate liquid phase formation, among other factors, play important roles in the graphitization process. Molten eutectic iron carbides solubilize disordered carbon and precipitate graphite platelets of crystallite size comparable to commercial graphite. Softwood-derived biographite is of the highest quality and demonstrates excellent electrochemical performance as anode material in a lithium-ion coin cell with 89% capacity retention over 100 cycles and > 99% coulombic efficiency.


 Please wait while we load your content...
Please wait while we load your content...