“Revitalizing” degraded solid oxide fuel cells in sour fuels for bifunctional oxygen catalysis in zinc–air batteries†
Abstract
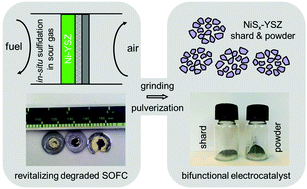
Electrochemical devices are central to sustainable energy storage and conversion. Currently, most of the spent devices end up in landfills or the conventional toxic and energy-intensive metal extraction processes. Advanced reuse and re-valorization practices are lacking due to both technical and economic barriers. To address this growing concern, we herein demonstrate that the fully degraded electrode of the state-of-the-art anode-supported solid oxide fuel cells (SOFCs) can be “revitalized” in situ during the operation. The facile treatment involves fueling the SOFC with the readily available fuels containing H2S and the electrode pulverization. The resulting powder comprises porous particles with mixed nickel sulfide nanoclusters dispersed on Y-stabilized ZrO2 (NiSx/YSZ) which enables superior activity in both the oxygen reduction reaction (ORR) and oxygen evolution reaction (OER). Its voltage gap between the ORR half-wave potential and OER potential at the benchmark 10 mA cm−2 reached 0.74 V, rendering stable and efficient performance in a rechargeable Zn–air battery. This work might boost the awareness of treating end-of-life electrochemical materials sustainably and pave new ways for the circular economy.
- This article is part of the themed collection: 2020 Green Chemistry Hot Articles


 Please wait while we load your content...
Please wait while we load your content...