Photocathode-assisted redox flow desalination†
Abstract
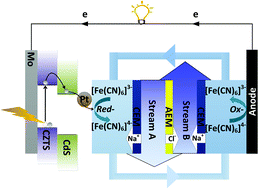
Desalination techniques, such as reverse osmosis, distillation, capacitive deionization, and battery desalination, require lots of electrical or thermal energy consumption. Herein, we propose a consumption-free electrochemical desalination method based on a light-driven photocathode with a Pt/CdS/Cu2ZnSnS4(CZTS)/Mo architecture. Modification of a CdS layer on CZTS can improve the desalination performance due to the formation of inner p–n junction between CdS and CZTS which enhances the separation of the photoexcited carriers without recombination. This photocathode-assisted electrodialysis desalination plays the dual functions of both energy conversion and ion removal with the blocking of ion exchange membranes. The [Fe(CN)6]3−/4− redox couples are recirculated between the anode and photo-cathode as the electrolyte while the salt streams are fed into the middle compartment. Under light illumination, this architecture produces photo-generated electrons to the redox couples with the conversion of [Fe(CN)6]3− to [Fe(CN)6]4− at the positive chamber, causing cation capture in the presence of an ion-exchange membrane. At the same time, [Fe(CN)6]4− is oxidized at the negative reservoir. The light-driven electrochemical reaction of electrolyte redox couples can result in a continuous desalination process. This work will be significant for consumption-free photoelectrochemical desalination research.


 Please wait while we load your content...
Please wait while we load your content...