Eco-friendly upconversion of limestone into value-added calcium formate†
Abstract
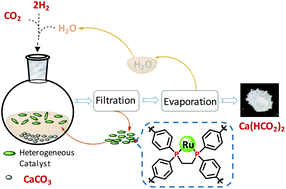
Although CaCO3 is a naturally available, inexpensive, abundant and extensively used chemical in various industries, to date, most of its conversions liberate CO2 into the atmosphere, which accounts for about 5–6% of global anthropogenic CO2 emission. Herein, the first synthesis of calcium formate (CF) by hydrogenation of an aqueous mixture of CaCO3 and CO2 using a heterogeneous catalyst in an unconventional CO2 reducing manner is reported. The phosphine-based heterogenized Ru catalyst efficiently converts CaCO3 into CF with good selectivity (>99%) and is readily separated from the reaction product, resulting in promising recycling ability. Thus, this work provides a promising strategy for the design of green and sustainable conversion of carbonate minerals in industry.


 Please wait while we load your content...
Please wait while we load your content...