Efficient catalytic conversion of cellulose to levulinic acid in the biphasic system of molten salt hydrate and methyl isobutyl ketone
Abstract
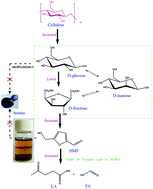
An efficient approach for the synthesis of levulinic acid (LA) by converting cellulose in a biphasic system consisting of molten salt hydrate (MSH), (LiCl·3H2O) and methyl isobutyl ketone (MIBK) using NbOPO4/HZSM-5 as the catalyst was reported. A series of molten salt hydrates (LiBr·3H2O, LiCl·3H2O, ZnCl2·3H2O, CaCl2·3H2O and Ca(NO3)2·3H2O) were evaluated in terms of their cellulose dissolvability and catalytic activity without any additional catalyst in the conversion of cellulose to LA. Among them, LiBr·3H2O, LiCl·3H2O and ZnCl2·3H2O were capable of dissolving and swelling cellulose and demonstrated excellent performance in cellulose conversion to LA even without any additional catalyst. High temperature favors the formation of LA while low temperature is beneficial to saccharides and 5-hydroxymethylfurfural (HMF). Using NbOPO4/HZSM-5, an efficient conversion of cellulose to LA was achieved in a biphasic system, affording a LA yield as high as 94.0%. The partition coefficient of HMF between the MIBK and LiCl·3H2O phases increased notably when the temperature was increased, proving that a biphasic solvent restrained the secondary reaction of HMF to humins. The reaction mechanism study based on the investigation of converting glucose, fructose and HMF to LA found that the side reactions of saccharides to humins are the key factors affecting the selectivity of LA in the investigated reaction system. Therefore, it is crucial to match the cellulose hydrolysis reaction with the subsequent reactions to maintain saccharides including glucose and fructose at low concentrations for highly efficient conversion of cellulose to LA.


 Please wait while we load your content...
Please wait while we load your content...