Electrochemical oxidative C(sp3)–H azolation of lactams under mild conditions†
Abstract
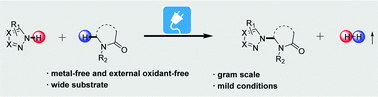
Lactam-containing structural compounds are ubiquitous in drugs and biomolecules. An electrochemical oxidative direct C(sp3)–H azolation of lactams has been reported under metal catalyst-free and external chemical oxidant-free conditions. This electrochemical C(sp3)–H/N–H coupling is characterized by its broad substrate scope of azoles and lactams under mild conditions at room temperature. Mechanistic studies suggested that the reaction possibly involves a radical process. Moreover, the site selectivity can be explained by DFT calculations. More meaningfully, a gram-scale synthesis method of flow electrochemistry was employed to show the scaled-up applicability of this transformation.


 Please wait while we load your content...
Please wait while we load your content...