Recycling lithium-ion batteries: adding value with multiple lives†
Abstract
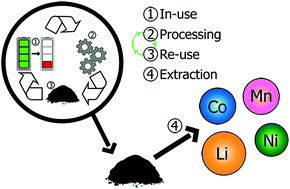
Resource extraction and limitations, valuable elements/minerals loss to land-fill, lack of recycling are growing concerns with increasing lithium-ion battery uptake and the synthesis of new cathodes (and more widely completely new materials for each battery). Factors such as these are exasperated in lithium-ion batteries which are currently one of the most expensive batteries to purchase and arguably are one of the most difficult to recycle efficiently. Herein, a mixed metal compound from the separation and processing of spent commercial batteries is compositionally, structurally, and electrochemically characterised as-received and sintered at temperatures up to 1000 °C. Whilst structural characterisation shows the significant presence of typical commercial layered cathode materials and their constituent elements via elemental analysis, this work shows that, without any further modifications to the as-received material, it is capable of delivering reversible capacities of over 300 mA h g−1 at 30 mA g−1 after 100 cycles as an anode in a “second-use” or “second-life” lithium-ion battery. Furthermore, the “second-life” lithium-ion battery has an increased intrinsic elemental value due to higher than normal concentrations of valuable metals (both in the fresh cathode and second-life anode) which can be extracted in future. This can be considered as an alternative approach to lithium-ion battery recycling.
- This article is part of the themed collection: 2020 Green Chemistry Hot Articles


 Please wait while we load your content...
Please wait while we load your content...