Insights into pressure tunable reaction rates for electrochemical reduction of CO2 in organic electrolytes†
Abstract
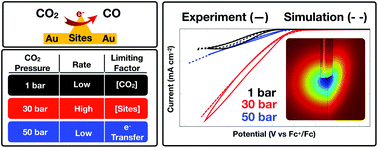
Electrochemical CO2 reduction rates are limited in aqueous-based electrolytes by the low solubility of CO2. We recently demonstrated that organic solvent-based CO2 Expanded Electrolytes (CXEs) can solubilize multi-molar amounts of CO2 at moderate pressures while retaining sufficient supporting electrolyte to facilitate electrochemistry. Up to an order of magnitude enhancement in CO2 reduction rates to CO was achieved on polycrystalline Au electrodes at faradaic efficiencies approaching 80%. Herein, we show that similar enhancements are observed on Cu catalysts as well, on the basis of enhanced current flow. On both systems, a maximum in CO2 reduction rate was observed at ca. 5 M CO2 concentration (i.e., 3.1 MPa head-space pressure) beyond which the rate decreases. To explain this counterintuitive phenomenon, we developed a detailed COMSOL-based mechanistic model of CO2 reduction in organic electrolytes under elevated pressures on Au electrodes. This model incorporates the dissimilar variations of the key physicochemical properties (viz., CO2 concentration, CO2 diffusion rate, and solution polarity) with CO2 pressure. We thereby demonstrate that the overall rate is limited by CO2 concentrations at lower than optimum CO2 pressures. At pressures higher than the optimum the rate is limited by both an attenuation of the first electron-transfer step and an increase in the ohmic resistance of the system. Excellent quantitative match between experimental and model-predicted rates lend credence to the proposed underlying mechanism of electrocatalytic CO2 reduction in CXEs. These fundamental insights provide guidance for the rational design and scaleup of highly efficient CXE-based CO2 reduction systems.


 Please wait while we load your content...
Please wait while we load your content...