In-depth interpretation of the structural changes of lignin and formation of diketones during acidic deep eutectic solvent pretreatment†
Abstract
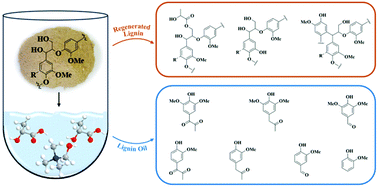
Deep eutectic solvents (DESs) as novel and green solvents can extract high-purity lignin from lignocellulose in a high yield; however, further utilization of the extracted lignin in the DES is a severe challenge in biorefinery processes. Nevertheless, the potential of lignin valorization can be precisely reflected by unveiling the chemical transformations of lignin during DES pretreatment. In this study, we aimed to understand the possible transformation pathway via thorough characterization of the regenerated lignin and lignin oil and transformation of lignin into value-added products. During this process, alkali lignin (AL) isolated from poplar was pretreated with choline chloride-lactic acid (ChCl-LA) and choline chloride-oxalic acid (ChCl-OA) at 80–120 °C for 6 hours to produce the regenerated lignin and lignin oil. The yield of the regenerated lignin ranged from 44 to 75% after DES pretreatment. During ChCl-LA pretreatment, γ-acetylated groups in the regenerated lignin were observed at low temperatures and then deacylation occurred at high temperatures, which was beneficial for oxidizing lignin and producing phenolic diketones. Additionally, the depolymerization of lignin fractions was the dominant reaction during DES pretreatment, accompanied by partial condensation reactions. More importantly, this is the first report of obtaining S- and G-derived diketones from lignin oil after DES pretreatment. The degradation mechanism of lignin in the DES system has been proposed. This work opens a way to produce value-added chemicals from lignin in DESs, which needs to be researched in the future.


 Please wait while we load your content...
Please wait while we load your content...