Development of an efficient and sustainable synthesis of 2-(3-methyl-1H-1,2,4-triazol-1-yl) acetic acid under continuous-flow conditions†
Abstract
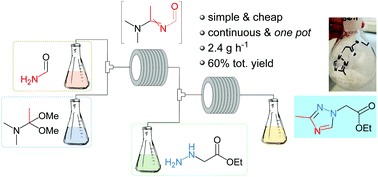
A novel, metal-free process for the challenging synthesis of 2-(3-methyl-1H-1,2,4-triazol-1-yl) acetic acid is reported, which features an efficient construction of the triazole ring under flow conditions. This continuous, one-pot method is atom economical, highly selective and environmentally benign, due to the avoidance of chromatography and isolation steps. Compared to the earlier batch routes, higher yields were achieved in a flow reactor. In addition, a highly-energetic intermediate could be controlled and handled in a safe manner. To generalize the established route, several triazoles were prepared under the described conditions, confirming that the methodology could find useful applications for the rapid and sustainable construction of differentially-functionalized 1,2,4-triazoles.


 Please wait while we load your content...
Please wait while we load your content...