Successful development of a method for the incorporation of Fmoc-Arg(Pbf)-OH in solid-phase peptide synthesis using N-butylpyrrolidinone (NBP) as solvent†
Abstract
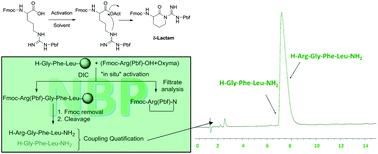
NBP has proved an excellent alternative solvent to the hazardous DMF for SPPS. Here we studied the incorporation of Fmoc-Arg(Pbf)-OH, one of the most problematic amino acids, into a growing peptide chain. The poor performance of this amino acid is attributed to the formation of a fully inactive δ-lactam, which causes a reduction in yield and very often the concomitant formation of the corresponding des-Arg peptides. This problem is exacerbated when NBP is used as solvent, presumably because of its high viscosity, which impairs the penetration of the coupling cocktail into the resin. To tackle this issue, we propose the following strategy for the safe introduction of Fmoc-Arg(Pbf)-OH in SPPS at 45 °C, keeping excesses to a minimum: 1.75 equiv. of the protected amino acids, 1.8 equiv. of DIC, and 1.5 equiv. of OxymaPure. The cornerstone of the strategy is to carry out in situ activation. In this regard, Fmoc-Arg(Pbf)-OH and OxymaPure dissolved in NBP were added to peptidyl-resin, allowed to reach the 45 °C, then half the DIC was added and left for 30 min, followed by the other half and some extra Fmoc-Arg(Pbf)-OH. During the entire process, the temperature was kept at 45 °C, with the double purpose of reducing the viscosity of NBP, thus facilitating the penetration of the coupling cocktail into the resin, and speeding up the coupling itself. It is envisaged that this strategy could be widely used to improve the performance of SPPS, including the industrial preparation of peptides using this approach.


 Please wait while we load your content...
Please wait while we load your content...