A solvent-free strategy to realize the substitution of I− for IO3− in a BiOIO3 photocatalyst with an opposite charge transfer path†
Abstract
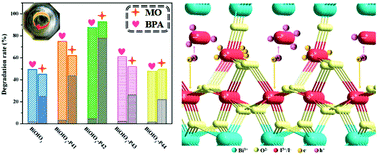
Substitutional doping is an effective way to improve photocatalytic activity, but substituting an individual ion for an atomic group has always been difficult to achieve. Herein, a simple in situ solvent-free strategy is reported for the synthesis of a photocatalyst by replacing IO3− in BiOIO3 with I− (BiOIO3-P42) via regulating the oxidation–reduction environment, which has been realized by changing the viscosity of the reaction system. Exhilaratingly, the change in the viscosity of the reaction system facilitated the phase composition change from pure BiOIO3 to I−-doped BiOIO3 and then to pure BiOIO3, which could be attributed to the synergistic effect of acidity and reducibility for various viscosities. This interesting phenomenon has been identified by XRD, XPS, SEM and HRTEM. Benefiting from the different valence states of iodine in I− and IO3−, the photogenerated electrons and holes could move in opposite directions due to the driving force provided by electrostatic attraction. I−-doped BiOIO3 therefore demonstrates an optimal photocatalytic removal rate in the degradation of methyl orange and bisphenol A, which is about two times higher than that of pure BiOIO3. We anticipate that such an in situ substitutional doping technique can be further investigated as a convenient method to design nanomaterials with boosted photocatalytic performance.


 Please wait while we load your content...
Please wait while we load your content...